Active Thermitic Material Discovered in Dust from the 9/11 World Trade Center Catastrophe
by Niels H. Harrit, Jeffrey Farrer, Steven E. Jones, Kevin R. Ryan, Frank M. Legge,
Daniel Farnsworth, Gregg Roberts, James R. Gourley and Bradley R. Larsen
The Open Chemical Physics Journal, Volume 2, pp. 7-31; 2009
Discussed at
JREF 1
Particularly
Post 292 (Red layer is Paint with Kaolin pigments),
Post 299 (grey layer is Micaceous Iron Oxide, used in anti-corrosion primer)
http://11-settembre.blogspot.com/2009/04/active-thermitic-material-claimed-in.html
A
French paper by Jérôme Quirant (Moorea34)
Some physical and chemical properties of thermite and thermate are discussed
here.
Note on page numbers: Each page of the PFD document has two page numbers: One written on top of the page, and the page number count of the PFD file. I will refer to pages by dual numbers as, for example "16(22)", meaning "page 16 of the PFD file (page 22 as written on top of page)".
Abstract
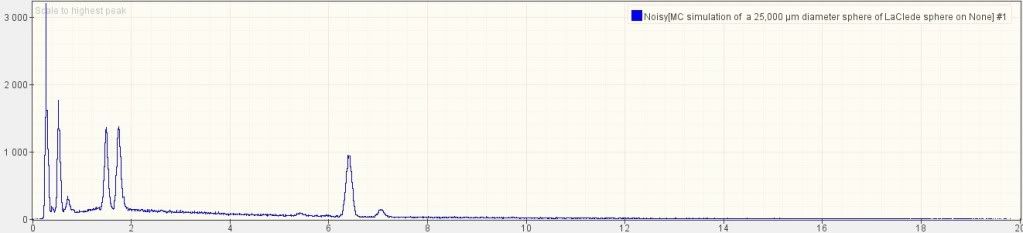
We have discovered distinctive red/gray chips in all the samples we have
studied of the dust produced by the destruction of the World Trade
Center. Examination of four of these samples, collected from separate
sites, is reported in this paper. These red/gray chips show marked
similarities in all four samples. One sample was collected by a
Manhattan resident about ten minutes after the collapse of the second
WTC Tower, two the next day, and a fourth about a week later. The
properties of these chips were analyzed using optical microscopy,
scanning electron microscopy (SEM), X-ray energy dispersive spectroscopy
(XEDS), and differential scanning calorimetry (DSC). The red material
contains grains approximately 100 nm across which are largely iron
oxide, while aluminum is contained in tiny plate-like structures.
Separation of components using methyl ethyl ketone demonstrated that
elemental aluminum is present. The iron oxide and aluminum are
intimately mixed in the red material. When ignited in a DSC device the
chips exhibit large but narrow exotherms occurring at approximately 430
°C, far below the normal ignition temperature for conventional thermite.
Numerous iron-rich spheres are clearly observed in the residue
following the ignition of these peculiar red/gray chips. The red portion
of these chips is found to be an unreacted thermitic material and
highly energetic.
The authors
- Niels H. Harrit: Associate Professor Emeritus, Ph.D. Chemistry, University of Copenhagen, 1975, Thesis: mechanistic photochemistry; has published on some nano-stuff, but his main method is Spectrophotometry, and his main research interests focus around organic chemistry, photochemistry, fluorescence. Profile at AE911T
- Jeffrey K. Farrer: PhD in Material Scince. Lab manager (read: technician) for electromicroscopy at BYU. Non-facult. Has about 4 legit scientific paper to his credit (3 at the Journal of Materials Science). Profile at AE911T
- Steven E. Jones: Former Physics professor at BYU. The driving force behind this effort. Profile at AE911T
- Kevin R. Ryan: B.S. Chem; Chemist, former Underwriters Laboratories manager (read: technician). Has never published at any science jurnal, except on 9/11 CTs. Profile at AE911T
- Frank M. Legge: Diploma of Agriculture; Logical Systems Consulting, Perth, Western Australia (Chemistry). Has a US Patent (4,765,309 of 08/23/1988) on a tracking device for solar panels. A Frank M. Legge has 1 paper on fatty diet. No other scientific credentials can be found. Profile at AE911T
- Daniel Farnsworth: (Grad?) student at Department of Physics and Astronomy, BYU. Did not sign AE911T petition.
- Gregg Roberts: Business Analyst, Technical Writer/Editor, VBA Dev, Psych. B.A., UT Austin, 1990; Graduate Work in Social Work. Gregg Roberts at AE911T
- James R. Gourley: Did not sign AE911T petition.
- Bradley R. Larsen: MS Geology, University of Utah; mineralogist for oil-gas and gold exploration for S&J Scientific Co.; Profile at AE911T
Criticism
The following weaknesses are found in the paper
- No proper peer-review
- Uncertainty about the source and chain of custody of the samples
- No comparison with thermite
- Comparison with one, unkown paint only is inconclusive
- Presence of C in all samples precludes DSC tests in atmosphere
- Energy release of >3.9J/g is proof that materials other than thermite react at 430°C
- The ignition point of 430°C is not shown to be indicative of any known thermite preparation and is contriaditcted by all known thermite preparations, which typically ignite above 900°C
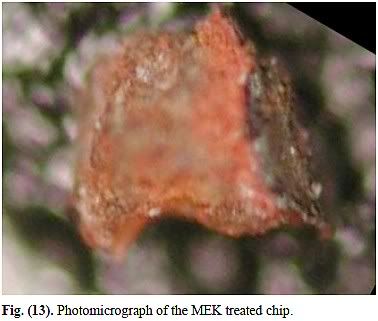
- The chip that was soaked in MEK has different spectrum from chips a-d
- It is not clear how elemental Al was identified in chip e
- Elemental Al is known to react with MEK, making it highly unlikely that elemental Al was found after soaking 55 hours in MEK
- Comparison with images, formulae and spectra of known Kalolin preparations for paint show striking similarities with red layer
- Comparison with images, formulae and spectra of known Micaceous Iron Oxide preparations show stiking similarities with grey layer
Peer-reviewed?
The authors, and their supporter and parroters, do not tire of pointing out that the paper had been subjected to a proper peer-review process when it was published at Bentham Open. But is this really so? Let's first look at how peer-review is usually done:
A scientific journal has an editor-in-chief, and possibly more editors
A paper submitted to a journal is first reviewed by the editor. The editor determines the subject matter, picks a few scientists whom she or he deems experts on the subject matter ("peers"), and sends them the submission for review
The peers send back a critique, with recommendations that can be of the following nature: i.) accept without reservations ii.) accept with minor corrections iii.) reject, and ask author to resubmit when certain problems are fixed, iv.) reject right away (paper has no chance)
The editor reads the peer-reviews, and then decides if and how to publish the paper
So the role of the editor is absolutely central to the peer-review process.
Bentham's "The Open Chemical Physics Journal" isn't exactly hustling and bustling and overburdening the editor-in-chief with hundreds of submissions to be processed. In fact, in all of 2009, the journal published only 4 papers (
View journal articles), totalling 46 pages.
How was the paper handled by TOCPJ's editor in chief? Editor in chief was
Professor Marie-Paule Pileni. When the Danish science magazine videnskab.dk contacted Mrs. Pileni to get further information about the recently published Harrit-paper, they were
in for a surprise (
Translation): Mrs. Pileni, the editor in chief, did not know that this paper had been published in her journal! She resigned immediately from her position at the Journal.
A paper that has been published without the knowledge of the editor in chief has
not gone through a proper peer review process.
(Further notes: The journal has a whopping 95 people on the
Editorial Advisory Board - that is more than 2 advisors per page published in the whole of 2009! TOCPJ has not a single paper published in 2010.
One has to wonder if the authors have tried to publish their paper at any journal that is not as obscure as TOCPJ. They would have saved money, because at TOCPJ, authors have to pay US$ 600 before their submissions are published!
And finally, a post about the reputation of Bentham OA journals in general at
JREF)
Comparison with paint.
Harrit ed.al. conclude that the red-grey chips don't come from paint. They determined this by running the same tests on a chip sample, and on paint samples:
Page 11(17): "
The initial objective was to compare the behavior of the red layer with paint when soaked in a strong organic solvent known to soften and dissolve paint. ... In marked contrast, paint chips softened and partly dissolved when similarly soaked in MEK."
Page 16(22): "
Several paint samples were also tested and in each case, the paint sample was immediately reduced to fragile ashes by the hot flame. This was not the case, however, with any of the red/gray chips from the World Trade Center dust."
Page 21(27): "
We measured the resistivity of the red material ... and obtained a rough value of approximately 10 ohm-m. This is several orders of magnitude less than paint coatings we found tabulated which are typically over 1010 ohm-m [31]. ... On the other hand, paint samples in the same exposure to MEK solvent became limp and showed significant dissolution, as expected since MEK is a paint solvent."
It is striking that they mention paint samples several times, did work on the,, but fail to say which paint they tested! There must be hundreds of thousands of different paints and primers on the market, thousands used in the WTC and on all the objects in it, and several that had widespread use in the construction of the towers. Whatever paint they tested, one can't generalize fron these results:
- Some paints will get partly dissolved in MEK, but not all
- Some paints will immediately burn to fragile ashes in hot flame, but not all
- Some paints have high elecrical resistance, but not all
It bears noticing that both elemental aluminium and Fe<sub>2</sub>O<sub>3</sub> (rust) are very usual ingredients of many paints.
Even more interesting is the authors' description of the red-grey chips as fitting very nicely a description of paint films: On page 20(26): "
Thus, the energetic nano-composite can be sprayed or even “painted” onto surfaces, effectively forming an energetic or even explosive paint. The red chips we found in the WTC dust conform to their description of “thin films” of “hybrid inorganic/organic energetic nanocomposite”. Indeed, the descriptive terms “energetic coating” and “nice adherent film” fit very well with our observations of the red-chips which survived the WTC destruction."
In typical "truther" parlance one could say: "The red-grey chips
look just like paint"
I will further note in passing that thermitic materials, if "painted" on anything - that is: applied as a very thin layer - would bring so little chemical energy to a surface unit as to render it pathetically useless as an incendiary. The red layers are about 15 microns thick in some samples. It can be shown that one needs 3 volume units of thermite to melt 1 volume unit of iron, so a 15 micron layer of thermite could at most melt 5 microns of steel from a steel element. That is pathetically little! 15 microns thermite on a surface of 1m<sup>2</sup> is a volume of 15cm<sup>3</sup> and a mass of about 60g, containing about 240kJ of energy. Not even enough to heat 1 bottle (750ml) of water from room temperature to boiling point.
Ignition of thermitic material
From
Thermite (Wikipedia):
"
Ignition of a thermite reaction normally requires only a simple child's sparkler or easily obtainable magnesium ribbon, but may require persistent efforts, as ignition can be unreliable and unpredictable. ... Even when the thermite is hot enough to glow bright red, it will not
ignite as it must be at or near white-hot to initiate the reaction. ... The reaction between potassium permanganate and glycerol or ethylene glycol is used as an alternative to the magnesium method. ... However, this method can also be unreliable and the delay between mixing
and ignition can vary greatly due to factors such as particle size and
ambient temperature."
Harrit ed.al. however claim to have ignited the red-grey chips, and started a thermitic reaction, at only 430°C: "
As measured using DSC, the material ignites and reacts vigorously at a temperature of approximately 430 °C, with a rather narrow exotherm, matching fairly closely an independent observation on a known super-thermite sample. The low temperature of ignition and the presence of iron oxide grains less than 120 nm show that the material is not conventional thermite (which ignites at temperatures above 900 °C) but very likely a form of super-thermite."
Did they verify that superthermite has this property of igniting at only 430°C? No. On page 17(23) they state: "
We would like to make detailed comparisons of the red chips with known super-thermite composites, along with comparisons of the products following ignition, but there are many forms of this high-tech thermite, and this comparison must wait for a future study.". They reference the following papers in conjunction with superthermite:
[20] http://www.p2pays.org/ref/34/33115.pdf
[21] http://www.mrs.org/s_mrs/sec_subscribe.asp?CID=2642&DID=115856&action=detail
[22] http://www.mrs.org/s_mrs/sec_subscribe.asp?CID=2642&DID=115976&action=detail
[30] http://awards.lanl.gov/PDFfiles/Super-Thermite_Electric_Matches_2003.pdf
The first 3 don't mention temperature of ignition. [30] explicitly states that super-thermite matches are particularly heat-resistant! See page 21(27):
"
The Super-Thermite electric matches produce no toxic lead smoke and are safer to use because they resist friction, impact, heat, and static discharge through the composition, thereby minimizing accidental ignition."
We must conclude then that Harrit ed.al.'s assumption that a low ignition temperature may be indicative of superthermite is inproven at best. It isn't even specuation, as they give two separate reasons in the paper why we should believe thermite and superthermite only ignite at very high temps above 900°C. It is best characterized as wishful thinking. In the worst case, the assertion is outright fraudulent. The statement "
the material ignites and reacts vigorously at a temperature of approximately 430 °C, with a rather narrow exotherm, matching fairly closely an independent observation on a known super-thermite sample" is
wrong. They did
not have a known super-thermite sample that ignited at 430°C. The statement, on page 15(21) "
That thermitic reactions from the red/gray chips have indeed occurred in the DSC (rising temperature method of ignition) is confirmed by the combined observation of 1) highly energetic reactions occurring at approximately 430 °C" is
wrong. As neither their own experiments nor the referenced literature indicate that any thermite reacts at 430°C, it is
not true that the "highly energetic reactions occurring at approximately 430C" confirms that these reactions were thermitic. It remains unkown.
Energy release
Several times in the paper, the authors label reactions or materials as "highly energetic": pp 1(7), 15(21), 19(25), 21(27), 23(29). This label is misleading.
The authors admit that the thermite they are looking at (Fe2O3+Al) releases a theoretical maximum of 3.9kJ of energy per gram (p21(27)). They are themselves struck by the observation that their red/gray chips sometimes release more energy than that, which absolutely proves that whatever burned there could not just be thermite! This is quickly explained away by assuming that the samples also contain organic substances. These must have be even more "highly energetic" than thermite. How "highly energetic" are organic compounds? Here are some examples (all values are kJ/g):
3.9: Thermite (Fe2O3 + Al)
4.6: TNT
7: strongest high explosives
8: Household waste
17: Sugar
18: Wood
18: PVC
22: Potato chips
23. PET
26: Polyester (plastic)
32: black coal
38: Body fat
41: Polystrene (plastic)
43: Jet fuel
46: Polyethylene, Polypropylene (Plastics)
We find that pretty much
all organic materials around us are a lot more "highly energetic" than thermite!
Coming back to the finding that one sample released more energy than thermite could possibly contain. This shows clearly that the samples are "contaminated". We must assume it highly likely that all samples, including those that released less than 3.9kJ/g heat, were contaminated with unknown organic material. Which raises the question: Could it not be the organic component of the samples that ignites at 430°C? Fact is: Many organic materials ignite at temperatures ranging between 230°C and 500°C. This includes all sorts of resins on which paints and primers are based.
The ignition point, and the energy released, point to small amount of organic materials. They are both nit typical for any known thermite.
On to another topic: The paper is clear that whatever they are analysing there is not ordinary thermite: p. 19(25): "
All these data suggest that the thermitic material found in the WTC dust is a form of nanothermite, not ordinary (macro-) thermite." What properties does nanothemrite have, other than consisting of nano-sized particles? The paper quotes this:
"
... when the ingredients are ultrafine-grain and are intimately mixed, the mixture reacts very rapidly, even explosively [20]. Thus, there is a highly energetic form of thermite known as an energetic nanocomposite or “super-thermite ..."
This wording makes it appear as if the ultrafine grains and intimate mixing somehow make the mixture (even more) "highly energetic". This is not so. The energy content does not change because of the physical structure of the thermite. All that changes is the reaction rate, or energy release rate.
Conclusions
The paper lists 10 characteristics or observations, before coming to a conclusion. I will address each of the 10 observations:
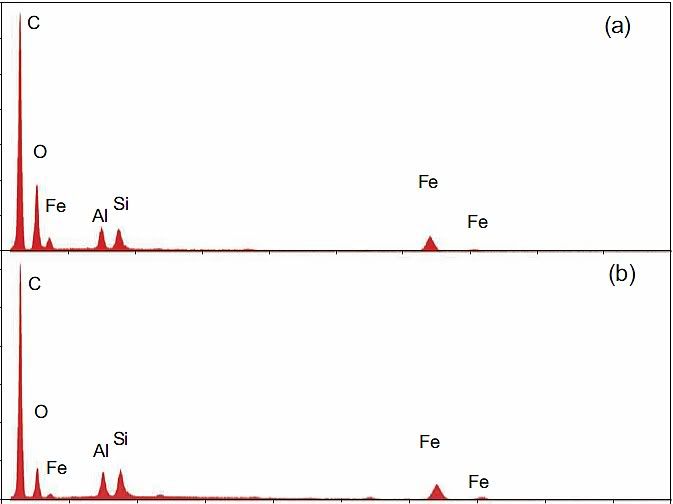
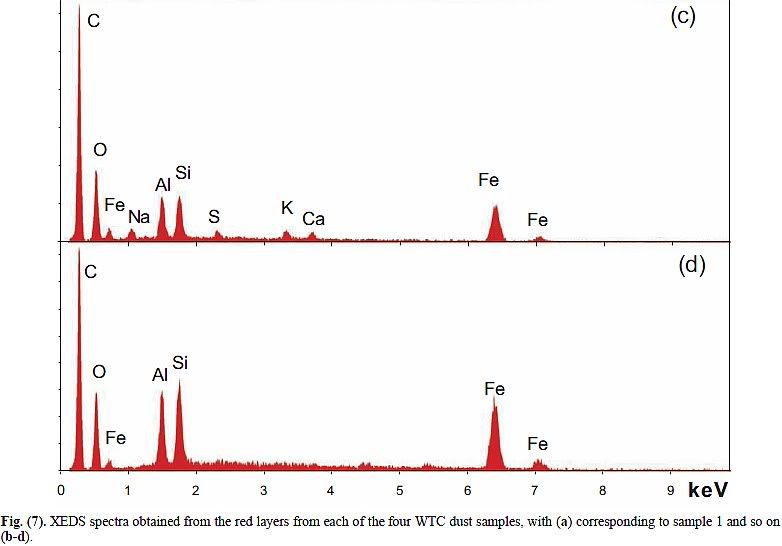
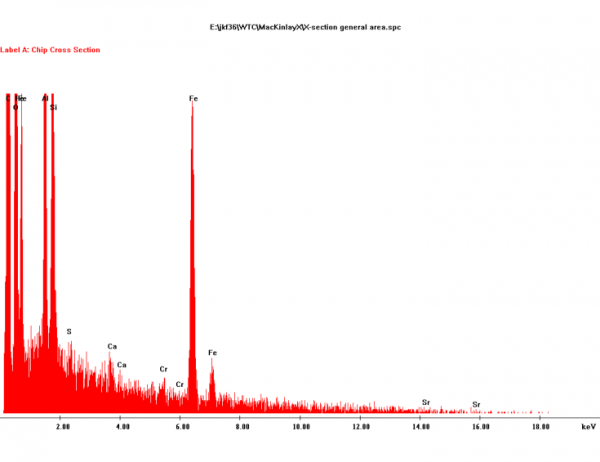
1. It is composed of aluminum, iron, oxygen, silicon and carbon. Lesser amounts of other potentially reactive elements are sometimes present, such as potassium, sulfur, lead, barium and copper.
They did not measure hydrogene, as that element is too light for their method (p.22(28): "
the middle-layer gray material contains carbon and oxygen and presumably also contains hydrogen, too light to be seen using this method". The elements listed first are precisely those that would be expected to be most abundand in the dust of a building collapse, as they are the constituents of the main materials: Steel Aluminium cladding, concrete and plastics. Sulfur, lead and copper are also abundand in office buildings. It is unclear why barium is mentioned in the conclusions - no data in the paper lists barium.
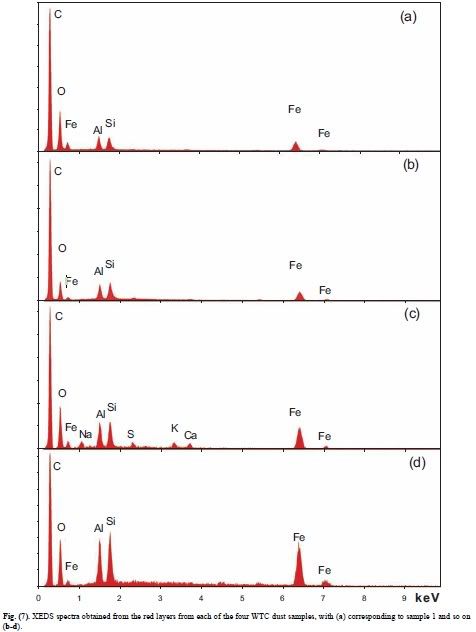
2. The primary elements (Al, Fe, O, Si, C) are typically all present in particles at the scale of tens to hundreds of nanometers, and detailed XEDS mapping shows intimate mixing.
This is primarily due to the authors choosing their samples precisely because of this property. They specifically searched for nano-sized constituents, and could be sure to find some in any dust.
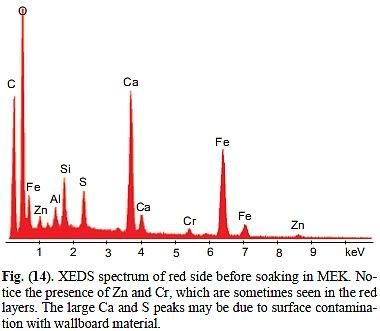
3. On treatment with methyl ethyl ketone solvent, some segregation of components occurred. Elemental aluminum became sufficiently concentrated to be clearly identified in the pre-ignition material.
It remains unclear what that segregation was supposed to achieve. But ok, we learn that aluminium was present. No surprise here.
4. Iron oxide appears in faceted grains roughly 100 nm across whereas the aluminum appears in thin platelike structures. The small size of the iron oxide particles qualifies the material to be characterized as nanothermite or super-thermite.
Actually, no. Conjecture. Nano-sized particles, incuding Fe2O3, are not at all unusual in paints.
5. Analysis shows that iron and oxygen are present in a ratio consistent with Fe2O3. The red material in all four WTC dust samples was similar in this way. Iron oxide was found in the pre-ignition material whereas elemental iron was not.
No one is surprised. Rust is plentiful in buildings, and a very ordinary constituent of anti-corrosion primers.
6. From the presence of elemental aluminum and iron oxide in the red material, we conclude that it contains the ingredients of thermite.
This is true. Fe2O3 and Al are indeed the main ingredients of thermite.
7. As measured using DSC, the material ignites and reacts vigorously at a temperature of approximately 430 °C, with a rather narrow exotherm, matching fairly closely an independent observation on a known super-thermite sample. The low temperature of ignition and the presence of iron oxide grains less than 120 nm show that the material is not conventional thermite (which ignites at temperatures above 900 °C) but very likely a form of super-thermite.
Not at all. There is no reference in the paper to that "independent observation on a known super-thermite sample". The paper itself references a paper that characterizes superthermite specifically as "heat resistant"!