Abstract
Harrit et al. [1] present 4 red-gray chips that they did a DSC test on. One of the chips was determined to yield a specific energy of 7.5 kJ/g. They believe that some of that energy yield comes from thermite in the red layer.
Assuming that the red layer contains at least as much Si as Al by weight and that Si is fully oxidized as SiO2, I have developed a simple spreadsheet to compute the minimum amount of energy that the organic matrix must contribute to raise the composite specific energy of the chip to the empirical value of 7.5 kJ/g. A number of cases is dicussed in which the following paramters are varied:
- Specific energy of thermite: Theoretical value of 3.96 kJ/g vs. a more realistic practical value of 3 kJ/g
- Mass of the gray layer: None vs. equal mass as red layer (this in effect doubles the effective specific energy of the red layer)
- Organic matrix: Values of 42 kJ/g (highest tabulated value of all organic polymers, for PP), 30 kJ/g (upper limit for most polymers), 20.4 kJ/g (epoxy) and 9.5 kJ/g (ideal polymer plus oxidizer composite) are considered.
Introduction
According to the data in [1] and Harrit et al.'s interpretation thereof, the red layer of their red-gray chips contains mainly just the elements C, O, Al, Si and Fe, while the gray layer is mainly iron oxide and chemically inert. They conclude that the red layer contains thermite (2 Al + Fe2O3) which reacts in the DSC test, and also an unidentified organic matrix that also reacts and is "itself energetic" (p. 28). Harrit et al. have heated four chips in a DSC up to 700 °C and measured heat flux. The most energetic of these four yielded a specific energy of 7.5 kJ/g (page 19):
Proceeding from the smallest to largest peaks, the yields are estimated to be approximately 1.5, 3, 6 and 7.5 kJ/g respectively. Variations in peak height as well as yield estimates are not surprising, since the mass used to determine the scale of the signal, shown in the DSC traces, included the mass of the gray layer. The gray layer was found to consist mostly of iron oxide so that it probably does not contribute to the exotherm, and yet this layer varies greatly in mass from chip to chip.
Later (p. 28) they correctly argue:
We observe that the total energy released from some of the red chips exceeds the theoretical limit for thermite alone (3.9 kJ/g). One possibility is that the organic material in the red layer is itself energetic.
When the chip as a whole exhibits 7.5 kJ/g, but it has constituent materials that are inert or have a lower energy density, then there have to be other constituents with an average specific energy that is significantly higher than 7.5 kJ/g. This already rules out conventional monomolecular explosives (Fig. 30 in [1]). So if the energy balance comes from the organic matrix, it has to react with an oxidizing agent: Either ambient oxygen, or, conceivably, some unidentified embedded oxidizer.
With reasonable assumptions to envelop ideal and realistic scenarios, it is possible to compute, dependent on hypothetical thermit contents, the minimum contribution of the organic matrix to the mass and energy yield of such a chip. I propose that, if significantly more of the energy yield comes from organic combustion than from thermite, then the characterization of the chips as "thermitic" as well as the interpretation of the DSC test results vis-a-vis nanothermite from literature are faulty.
Assumptions
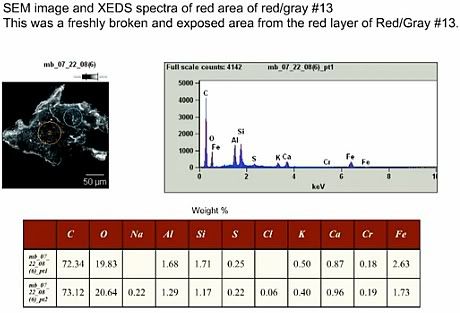
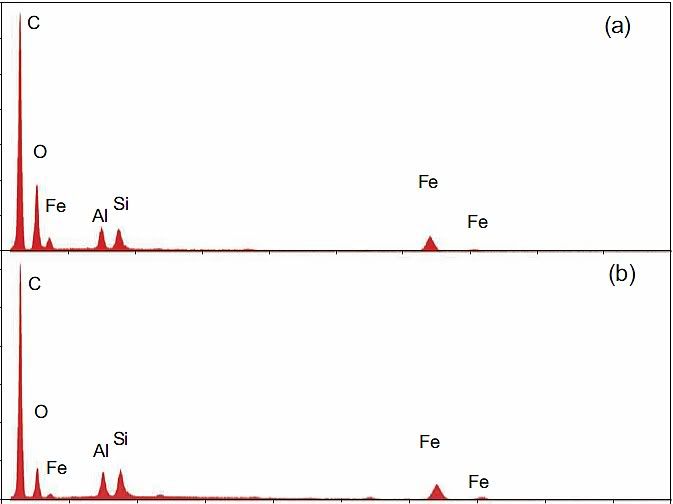
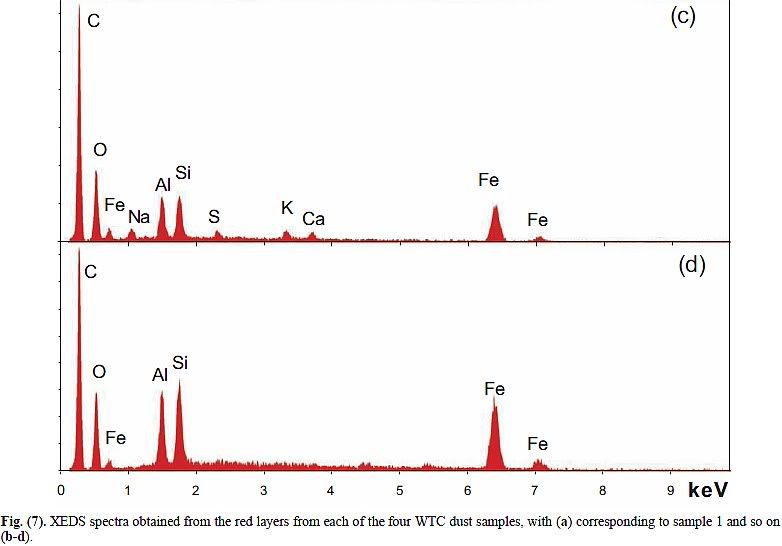
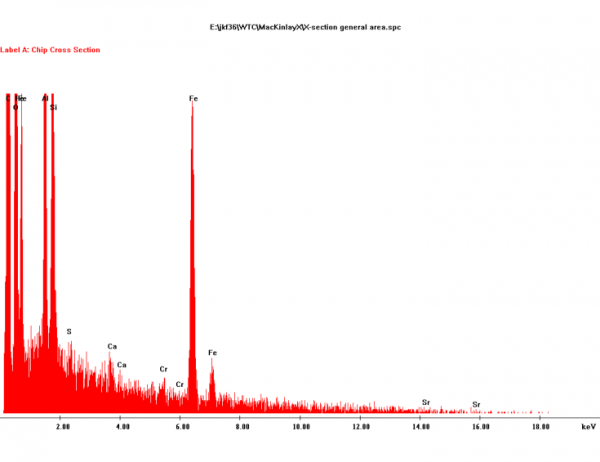
It is known from practically all EDS spectra of red layers or residues that Harrit et al. present that the silicon amount is at least equal to, and often exceeds, the aluminium content. This is easy to verify: In Fig. 7, and Fig. 11, the Al- and Si-peaks are about equal, in Fig. 14, 16, 18, 25 and 26, the Si-peak is significantly higher than the Al-peak. The only exception, Fig 17, is from a small spot location specifically chosen for its high Al-content. That chip however contains less Al overall than Si (Fig. 14). Judging from Fig. 16 I find that Si is accompanied by enough O to form silica.
I will therefore assume that, for all chips tested in the DSC,
- Si was present in the red layer in a mass fraction equal to that of Al.
- Si is fully oxidized as SiO2. Since Si and O appear in silica in a mass fraction of 28:(16+16), this means that for each mass unit of Al, there are 32/28 = 1.14 mass units of O.
These are the only assumptions that are not "thermite friendly", but they follow from the data! I have three more assumptions that remain constant throughout all cases and that are either "thermite friendly" (i.e. deviation from them would render the case for Harrit et al.' "thermite" hypothesis even more unrealistic), or almost neutral:
- I assume in all cases cases that all the Al is mixed with iron oxide as stoichiometric thermite: 2 Al + Fe2O3 (molar masses 2*26.98 + 159.69) consists of 74.7% iron oxide and 25.3% Al, so for each mass unit of thermite, there are 0.253 mass units of Al and 0.541 mass units of SiO2 (0.253 + 0.253*32/28).
- The balance of the mass of the red layer is assumed to be organic material, with or without embedded oxidizer.
- The gray layer is always present and assumed by Harrit et al. to not contribute to the exotherm. I disagree somewhat with this assumption - it is quite possible for hematite to experience exotherm phase changes when heated, but that will pale against redox reactions of fuel, so I ignore that and accept the assumption.
Parameters and cases
It is well known that many solid organic compounds, including many that could form such a matrix (epoxy, alkyd, other resins) are highly energetic. Practically all of them release more than the 3.96 kJ/g that thermite does. For non-halogenated polymers (Harrit et al. found no significant signals for halogenes), tabulated values for effective heat of combustion range from 12.0 kJ/ for Polyimide thermoplastic over 20.4 kJ/g for epoxy and around 25.5 kJ/g for Polyamides to 41.9 kJ/g for Polypropylene ([2], Table A-5). The large majority are in a range between 15 and 30 kJ/g. Itherefore propose that the effective specific energy of the unknown organic material cannot exceed 42 kJ/g, and probably does not in fact exceed 30 kJ/g. I will further consider the case that the matrix is epoxy, as Millette found chips with an epoxy matrix.
The previous paragraph considers organic combustion with oxygen from ambient air. Such a process limits the burn rate and would render the red layer material less-than-explosive. I will consider a hypothetical polymer readily mixed with pure oxygen as oxidizer: The highest energy yield is tabulated for Polypropylene (PP), at 42 kJ/g. PP has a sum formula of (C3H6)n. Complete combustion follows the formula: C3H6 + 4.5 O2 -> 3 CO2 + 3 H2O + heat. This means that a composite of PP and O2 would have to incorporate 144 (9*16) g of oxygene per 42 grams of PP. The effective specific energy of that composite would consequently drop to 42 kJ/g * 42/(42+144) ~ 9.5 kJ/g. Of course any actual solid oxidizer (such as perchlorates or permagnanates) contains additional mass that would further decreases the effective specific energy. I will use 9.5 kJ/ as an upper limit to envelop any and all (organic polymer + oxidizer) composites.
I will assume in the ideal case that all the Al is present as metal, none is oxidized, and that it reacts perfectly with all the available iron oxide to release the theoretical maximum of 3.96 kJ/g. In a more realistic case, I will assume the same proportions of aluminium and iron oxide, but consider that a sgnificant proportion of nano-Al is passivated, or that it won't react to completion, such that the effective energy yield of the thermite is decreased to 3 kJ/g.
The mass proportions of red:gray layers isn't known and difficult to estimate. However, as the gray layer is mostly iron oxide (>5 kg/L) and the red layer is only partly iron oxide, all other constituents lighter (Al: 2.7 kg/L; silica: 2.65 kg/L; polymers: usually <2 kg/L), it is clear that the gray layer has a significantly higher density. Harrit suggest that both layers are usually of similar thickness. In my unrealistic cases, I will ignore the gray layer, or assume its mass is 0, in the realistic cases I'll assume both layers have the same mass. That effectively doubles the specific energy of the red layer, from 7.5 to 15 kJ/g.
Calculations
Formulas
I created a spreadsheet with five input cells (numbers (1), (2), (6), (7) and (9) below) and with six relevant output cells (numbers (4), (5), (11), (13), (14), (15) and (17) below). The formulas provide a generic description of the spreadsheet formulas, so you can recreate the spreadsheet with any software you prefer:
(1) The mass of the red layer is constant: 100%.
(2) The mass proportion of thermite in the red layer, expressed in %
(3) The mass proportion of Al in the red layer: =(2)*0.253
(4) The mass proportion of SiO2 in the red layer: =(3)*60/28
(5) The mass proportion of the organix matrix is the balance: =(1)-(2)-(4)
(6) The specific energy of thermite: In the "ideal" case 3.96 kJ/g, in the "realistic" case 3 kJ/g
(7) The mass of the gray layer: In the "ideal" case 0, in the "realistic" case 100% (of the red layer)
(8) Total mass of the Chip: =(1)+(7)
(9) The measured specific energy of the chip, in kJ/g; constant at 7.5 kJ/g for the purpose of this article
(10) The specific energy of the red layer alone: =(9)*(8)/(1)
(11) The contribution of thermite to the specific energy of the red layer in kJ/g: =(6)*(2)
(12) The contribution of the organic matrix to the specific energy of the red layer in kJ/g: =(9)-(11)
(13) The specific energy of the organic matrix: =(12)/(5)
(14) The mass ratio of organics:thermite: =(5)/(2)
(15) The energy contribution ration of organics:thermite: =(12)/(11)
(16) The energy contribution of thermite, in % of total energy release: =(11)/(9)
(17) The energy contribution of organics, in % of total energy release: =(12)/(9)
For each of the three cases, I kept the specific energy of thermite (6) and the mass of the gray layer (7) constant and played with the mass proportion of thermite (2) such that the specific energy yield of the organic matrix (13) reached the target values of 9.5, 20.4, 30 and 42 kJ/g. I then copied the resulting values of interest into the tables.
Tabulated results
I have modeled three cases - one unrealistic, one half realistic, one realistic. In each case, I have tabulated results for 5 mass proportions of thermite:
- Thermite fixed at 12%. This means an Al-content just over 3%. I chose this value, as quantifications of XEDS spectra of red layers suggest there is less than 3% Al in them
- Thermite chosen such that the organic matrix computes to the specific energy yield of 9.50, which is a theoretical upper limit for organic polymer with embedded stoichiometric oxygen
- Thermite chosen such that the organic matrix computes to the specific energy yield of epoxy, 20.40
- Thermite chosen such that the organic matrix computes to the specific energy yield of 30 kJ/g, which is a realistic limit defined in my assumptions
- Thermite chosen such that the organic matrix computes to the specific energy yield of 42 kJ/g, which is the maximum for all organic polymers
Ideal case: Gray layer 0%, Thermite 3.96 kJ/g, chip yields 7.5 kJ/g
"Ideal" means "unrealistic" - this case gives absolute theoretical maxima for the contribution of thermite to the measured exotherm. It is however impossible to actually reach these values, as the energy yield of thermite in practice never reaches the theoretical maximum of 3.96 kJ/g, and there was in fact a gray layer that contributed significant mass but no significant energy.
| Mass of Thermite % of red layer (2) |
Mass of SiO2 % of red layer (4) |
Mass of Organics % of red layer (5) |
Contrib. Thermite kJ (11) |
Yield of Organics kJ/g (13) |
Mass Thmt:Poly (14) |
Energy Thmt:Poly (15) |
Contrib. Polymer % (17) |
| 12.00 | 5.69 | 82.31 | 0.48 | 8.53 | 1:6.86 | 1:14.78 | 93.66% |
| 19.90 | 9.44 | 70.66 | 0.79 | 9.50 | 1:3.55 | 1:8.52 | 89.49% |
| 49.39 | 23.43 | 27.18 | 1.96 | 20.40 | 1:0.55 | 1:2.83 | 73.92% |
| 55.87 | 26.50 | 17.63 | 2.21 | 30.00 | 1:0.32 | 1:2.39 | 70.50% |
| 59.52 | 28.23 | 12.25 | 2.36 | 42.00 | 1:0.21 | 1:2.18 | 68.57% |
More realistic case: Gray layer 0%, Thermite 3.00 kJ/g, chip yields 7.5 kJ/g
"More realistic" means "still unrealistic" - The thermite yield is now reasonable, but I still ignore the very real mass of the gray layer.
| Mass of Thermite % of red layer (2) |
Mass of SiO2 % of red layer (4) |
Mass of Organics % of red layer (5) |
Contrib. Thermite kJ (11) |
Yield of Organics kJ/g (13) |
Mass Thmt:Poly (14) |
Energy Thmt:Poly (15) |
Contrib. Polymer % (17) |
| 12.00 | 5.69 | 82.31 | 0.36 | 8.67 | 1:6.86 | 1:19.83 | 95.20% |
| 18.20 | 8.63 | 73.17 | 0.55 | 9.50 | 1:4.02 | 1:12.74 | 92.72% |
| 47.64 | 22.60 | 29.76 | 1.43 | 20.40 | 1:0.62 | 1:4.25 | 80.94% |
| 54.57 | 25.89 | 19.54 | 1.64 | 30.00 | 1:0.36 | 1:3.58 | 78.17% |
| 58.55 | 27.77 | 13.68 | 1.76 | 42.00 | 1:0.23 | 1:3.27 | 76.58% |
Realistic case: Gray layer 100%, Thermite 3.00 kJ/g, chip yields 7.5 kJ/g
"Realistic" means that all assumptions are now within the bounds of what is possible in practice - it does not mean that these are probable values! Al still ist best estimated as less than 3% of the mass of the red layer! This case merely provides realistic maxima of hypothetical thermite contribution to mass and energy yield af that chip and its 7.5 kJ/g.
| Mass of Thermite % of red layer (2) |
Mass of SiO2 % of red layer (4) |
Mass of Organics % of red layer (5) |
Contrib. Thermite kJ (11) |
Yield of Organics kJ/g (13) |
Mass Thmt:Poly (14) |
Energy Thmt:Poly (15) |
Contrib. Polymer % (17) |
| 12.00 | 5.69 | 82.31 | 0.36 | 17.79 | 1:6.86 | 1:40.67 | 97.60% |
| n/p | n/p | n/p | n/p | 9.50 | n/p | n/p | n/p |
| 19.95 | 9.46 | 70.59 | 0.60 | 20.40 | 1:3.54 | 1:24.06 | 96.01% |
| 36.38 | 17.26 | 46.36 | 1.09 | 30.00 | 1:1.27 | 1:12.74 | 92.72% |
| 45.82 | 21.74 | 32.44 | 1.37 | 42.00 | 1:0.71 | 1:9.91 | 90.84% |
Discussion
The first, fully unrealistic case, shows that even under the most thermite-friendly assumptions - the highest possible energy contribution of the organic matrix and ideal composition of thermite, with no losses, and neglected gray layer mass, the organic matrix provides more than twice as much energy as the hypothetical thermite. In the case of a more typical or expected epoxy matrix, this factor increases to almost 3. If XEDS readings are reliable in their indicating 3% of Al or less, then the organic matrix provides at least 93.66% of the measured energy, or almost 15 times as much as thermite.
When we consider that nanothermite can't in practice yield the theoretical maximum but will in practice be limited to perhaps 3 kJ/g, then even the best, most "thermite-friendly" organic matrix would contribute 76.58% of the measured energy, which is more than 3 times the energy that thermite contributes in that case - even when the gray layer is ignored. If the matrix is epoxy, then thermite would only contribute 21% of the energy. If the polymer carries its own oxidizer, then it must contribute more than 4 times the mass of thermite and more than 12.7 times more energy.
Putting the gray layer into the equation, the last remaining hope that thermite could play a significant role is shattered: The best realistic case, with an organic matrix that has 42 kJ/g, there could be almost 46% thermite in the red layer, but it would contribute only 11% of the energy. A more typical organic material with 30 kJ/g would have to outweigh the thermite by mass and yield 92.72% of the total energy. An epoxy matrix would have to outweigh thermite by a margin of 3.54:1, giving it 24 times the energy content of the 20% thermite. However, as there probably isn't actually more than 3% Al in the red layers, which means no more than 12% thermite, we find that 7.5 kJ/g for the entire chip means that 97,60% of its energy must come from organic combustion.
The spreadsheet shows that, as long as the gray layer has more than 26.5% of the mass of the red layer. it is not possible at all to reach 7.5 kJ/g with an ideal hypothetical (polymer+O) composite that is independent from ambient air, regardless of thermite content (nor can thermite, with its mere 3.96%, explain that value).
Since in all realistic scenarios, no more than 36% of the mass of the red layer would be thermite, and that thermite would be intimately mixed with silica and organic matrix in a nanocomposite with a very high surface-to-volume ratio, we have to assume that the heat released by both the thermite and the organic matrix would increase the temperature of all ingredients uniformly. A significant proportion of that heat is lost in gasification of the organic polymer. It seems unlikely that any part of such a mix could reach a temperature near the melting point of iron, as Harrit et al. seem to suggest (page 19).
Conclusions
With only two limiting assumption - that there is as much Si as Al, and that it is fully oxidized to silica - I have shown that the theoretical maximum contribution of thermite is under 1/3 of the energy yield of that chip. This result of just 31.43% energy from thermite holds true only for a carefully chosen but impossible set of conditions: Perfect efficiency of the thermite, no mass contribution from inert gray layer, and the most energetic solid organic polymer fuel.
Considering realistic parameters - that the hypothesized nanothermit will yield no more than 3 kJ/g (about 75% of the theoretical maximum) in practice, and that the gray layer has the same, but chemically inert, mass as the red layer, I find that the organic matrix must contribute at least ten times as much energy as thermite. This factor of ten holds true only with an ideal organic fuel. A realistically chosen organic fuel, with a specific energy between 20 and 30 kJ/g, would have to have 1.27 to 3.54 times the mass of thermite, and contribute 92.7 - 96.0% of the energy to boost that chip to its empirically determined 7.5 kJ/g. In these scenarios, the thermite content falls to 20%
Considering that the actual Al-content of the red layers is probably under 3%, I find that thermite can at most contribite 2.4% of the 7.5 kJ/g
It is not within the realm of the practically possible to hypothesize that the organic matrix itself contains an embedded oxidizer.
With those findings, this red-gray chip cannot reasonably be called "thermitic" and cannot be explosive.
References
[1] Harrit N. H.; Farrer, J.; Jones, S. E.; Ryan, K. R.; Legge, F. M.; Farnsworth, D.; Roberts, G.; Gourley, J. R.; and Larsen, B. R.: Active Thermitic Material Discovered in Dust from the 9/11 World Trade Center Catastrophe. The Open Chemical Physics Journal, 2009, 2, 7-31
[2] Lyon Richard E.; Janssens Marc L.: Polymer Flammability. May 2005 - Final Report for the U.S. Department of Transportation and FAA. Report No. DOT/FAA/AR-05/14