1. Abstract
Mark Basile has presented his analysis of red-gray chips he found in dust collected in lower Manhattan very shortly after the collapse of the World Trade towers on 2001/09/11 [1]. He concludes that his experiments confirm a similar but more comprehensive study published by Harrit e.al. [2]. Harrit e.al. have in turn accepted Basile's findings as confirmation of their conclusions: That the red-gray chips are thermitic in nature.
I will show that this conclusion is not warranted in any way. Instead, Basile's favorite specimen is organic by nature, with at most 1.3%, but perhaps 0%, of the heat of reaction coming from a thermite reaction, the balacnce, 98.7%-100%, from ordinary organic hydrocarbon combustion.
If this result is a “confirmation” of Harrit e.al., as 9/11 Truthers like to point out, then clearly this puts in grave doubt the affirmation that Harrit's chips were of thermitic nature.
2. Introduction
Mark Basile is a chemical engineer whose name is the second among those that Harrit e.al [2] thank for in their Acknowledgments (page 30). He held a videotaped presentation at the Porcupine Freedom Festival in Lancaster, New Hampshire on June 26th, 2010 at 4pm [1]. According to the presentation, between 30:00 and 31:26 minutes, he received a bag with “a few table-spoons” of dust collected by Janette MacKinlay in January 2008. Janette MacKinlay is also the contributor of dust sample 1 to Harrit e.al.
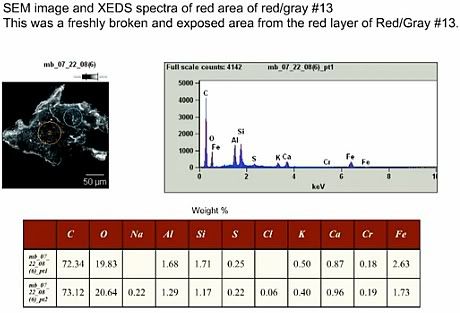
Basile isolated various dust particles from the sample, using a magnet and a petri dish, among them “iron based microspheres, Red/gray chips, Red chips, Rust, Wire”, also “Silicate or glassy spheres”. One particular red/gray flake, designated “#13” (he calls it “his lucky thirteen”), was photographed through a microscope, analyzed using XEDS and then heated till ignition, and the burning recorded on video through a microscope.
Basile found iron and aluminium atoms in the red layer, and concludes that the combustion he observed it probably thermitic.
3. The data
Here is the XEDS graph for chip # 13, shown at 39:30 in the video:
He explains that the table of weight-% values is derived from a standard software routine on the XEDS. I want to advise the reader to be careful with such derivations: The peak height or x-ray counts in XEDS spectra depend somewhat on a number of factors, such as surface and bulk geometry of the sample, and the presence or absence of materials that may tend to attenuate signals. The values aren't wrong, but remember that they come with a certain margin of error that is difficult to estimate.
Later in the presentation, between 41:43 and 42:00, Basile shows how such a red chip burns. I note his dramatic description of the event, but basically I just see something burning. So where does the heat of that reaction come from?
4. Discussion
Through most of this discussion, I will use the most “thermite-friendly” data and assumptions. By this I mean, I will take the data points where thermite ingredients were most abundant. I will assume that iron was indeed Fe2O3 and enough aluminium indeed elemental. I will assume that all of these intgredients contributed to a thermite reaction, with no losses and almost prefect execution. I will try to minimize the amount of hydrocarbons and its energy contribution so that the thermite reaction becomes as dominant as it can get.
4.1 Chemical composition - “thermite friendly”
The elemental composition that Basile shows in that table above translates into a mix of chemical compounds. Let's see what that mix looks like under the assumption that it contains the maximum amount of thermite, and minimum amount of other energetic compounds. To do so, I will use the weight-% figures in the first line, as the values for iron and aluminium, the main ingredients of thermite, are higher there.
Basile himself explains at 39:46:
In large part, it's an organic material of some sort
I agree fully: According to his quantification, more than 72% of the red layer are carbon. Basile knows that almost all of the carbon is bound with oxygen and hydrogen (and possibly other elements mixed in) to form hydrocarbons. This immediately means that more than 72% of the red layer is some kind hydrocarbon: Hydrocarbons also contain, as the name implies, hydrogen (H), which doesn't show up in an XEDS graph because it is too light. In many organic compounds, the molar (atom count) ratio of C:H is between 1:1 (for example Benzene, C6H6, which is a building block for many more complex molecules, including epoxies or TNT) and 1:2 (for example MEK, C4H8O, an organic solvant), which translates to mass ratios between carbon and hydrogen between 12:1 and 6:1. Staying on the careful (“thermite-friendly”) side, 72% of organic C in the red layer implies at least an addition of 72%/12 = 6% H by weight (increasing the sums of weights to 106%, if you will). Almost all hydrocarbons also have oxygen in their molecules, and certainly the nearly 20% of O are not bound to the metals.
Now let's try to use up as much of all the elements in inorganic compounds as we can – with the exception of aluminium, which I will assume to be totally elemental (an unrealistic assumption – Al is always oxidized on its surface):
All the silicon is fully oxidized as SiO2
All the iron is fully oxidized as Fe2O3
All the sulfur and some of the calcium is assumed to be contamination with gypsum: CaSO4.2H2O (this adds a tiny, almost negligible amount of H, which I do take into account)
All the chromium and some of the calcium is calcium chromate: CaCrO4
The remainder of calcium is calcium carbonate (this will remove a bit organic carbon): CaCO3
All the potassium is potassium carbonate (removes more C from organics): K2CO3
If you do that, you will find that of the 19.83% oxygen in Basile's table, only 4.86% can be accounted for by inorganic compounds, while almost 15% must be part of the hydrocarbon matrix; Not more than 3% of the carbon could be explained as inorganic (inert) carbonates of calcium and potassium. The hydrocarbon matrix would have C:O:H in ratios of about 12:3:1 by mass, or 5:1:5 by atom count – this assuming a hydrocarbon very poor in hydrogen. A C:O molar ration of 5:1 is not far from the 6:1 ratio that I computed for a typical cured epoxy (unpublished private work), but any commenter is invited to correct me on that point.
The most “thermite-friendly” composition of the red layer computes thus to, by weight:
87.8% hydrocarbon matrix
3.54% iron oxide (thermite ingredient)
1.58% aluminium (elemental, thermite ingredient)
7.08% other inorganic compounds
4.2 Stoichiometric thermite
The thermite reaction is
Fe2O3 + 2 Al → Al2O3 + 2 Fe
1 mol of Fe2O3 has a mass of 159.69 g, and 2 mols of Al have a mass of 53.96 g, so if you want to mix these two ingredients in ideal (what the chemist calls “stoichiometric”) proportions, you'd have to take 159.69 / (159.69+53.96) = 74.7% iron oxide, and (the balance of) 25.3% pure aluminium.
In Basile's red layer, these components appear 3.54 : 1.58, or 69% : 31%. So there is relatively too much Al – and indeed, I unrealistically assumed that all the Al would be elemental, when in fact at least some of the Al will always be oxidized, as the top few nanometers of all aluminium surfaces react with oxygen almost instantly. A few nanometers sounds like very little, but of course Basile, like Harrit e.al., claim we are dealing with nano-thermite, so a few nanometers is significant!
If you want to pair 3.54% by weight iron oxide stoichiometrically with Al, you need 3.54% * (74,7%/25.3%) = 1.20% aluminium, which means the mass fraction of ideal thermite in Basile's red layer is at most 3.54% + 1.20% = 4.74%.
4.3 Energy content of thermite and hydrocarbons
What's the maximum energy density of thermite? According to [2], page 28,
the theoretical limit for thermite alone [is] (3.9 kJ/g)
The value is actually a little closer to, but still slightly under, 4.0 kJ/g, but as no reaction actually reaches the theoretical upper limit, 3.9 is a good (and optimistic, i.e. “thermite-friendly”) value to go with.
As shown above, at most 4.74% of the red layer can consist of the thermite ingredients in perfect proportions, so this thermite would contribute only 3.9 kJ/g * 4.74% = 0.185 kJ/g of energy to the red layer, per mass of the same.
What's the energy density of the organic matrix? Since we don't know what hydrocarbon we are looking at, and since it is difficult to find tabulated values for energy density of organic polymers such as epoxies, I can only provide estimates. But it is well known that practically all hydrocarbons combust or degrade exothermally under air when heated sufficiently (after degradation,. Reactions will continue and can be quite complex). Wikipedia [3] lists a few organic materials and they energy density under air (the unit MJ/kg is the same as kJ/g, since 1 MJ = 1000 kJ, and 1 kg = 1000g):
46 kJ/g: Some plastics (Polypropylene, Polyethylene), petrol, Diesel fuel
37 kJ/g: Body fat:
26 kJ/g: Polyester
23 kJ/g: PET plastic
16-18 kJ/g: Carbohydrates (sugars, starch). Wood, PVC, proteins
15 kJ/g: Dry cowdung and cameldung
5 kJ/g: Teflon plastic
The low value is actually fluoropolymer, in which fluor dominates over hydrocarbon, and it is a flame retardant material. I find that practically all pure hydrocarbons have an energy density of 15 keV or more, sometimes much more. It is certainly reasonable to expect that the same is true for the hydrocarbon matrix of the red layer.
With the hydrocarbon constituting 87.8% of the red layer, would contribute 15 kJ/g * 87.8% = 13.17 kJ/g of energy to the red layer, per mass of the same.
All the other inorganic compounds have been assumed to already be fully oxidized, they are inert. These 7.08% of the red layer mass would contribute nothing to a combustion, or 0 kJ/g.
To sum up: hydrocarbons would contribute 13.17 kJ/g to the red layer, and thermite 0.185 kJ/g, in the most “thermite-friendly” case, for a sum of 13.36 kJ/g. Thermite contributes 1.4% to this heat, and hydrocarbons 98.6%. In other words, hydrocarbons provide more than 71 times reaction energy than thermite.
4.4 Less “thermite-friendly” assumptions
Each time I made assumptions, I chose the values such that thermites's relative contribution to the heat realease would be maximized. I chose...
the data set with the higher abundance of Fe and Al: Factor 1.5
the highest possible energy density of thermite: Factor 1.3
the lowest realistic energy density of hydrocarbon: Factor 1.2-1.7
In addition, I assumed that as much aluminium would be elemental as could be possibly oxidized by the available iron oxide. There is no reason to assume that any elemental Al would be present. Taking into account these factors, hydrocarbon heat release would dominate that of thermite more realistically by a factor (71 x 1.5 x 1.3 x 1.2) ~ 166. If there is any thermite at all, that is.
Here is how I derived these three factors:
4.4.1 Basile's second line
I used Basile's first estimate of elemental fractions, with 2.63% iron (3.54% iron oxide) as the limiting factor of thermite abundance. In his second line, there is only 1.73% iron, which, if completely oxidized, would be 2.33% iron oxide. Stoichiometric mix with 0.79% pure aluminium gives us 3.12% thermite – that's less than the first case by a factor of 1.5
4.4.2 Thermite never perfect
Even if you could mix thermite stoichiometrically, it would never react 100%. Certainy, a loss of at least 30% can be expected, especially since the aluminium- and iron oxide particles must be so few and far between in the matrix, at such low abundances (well under 5% each). This gives another factor of at least 1.3
4.4.3 Hydrocarbon more energetic
I chose an energy density value for the unknown hydrocarbon on the low end of the scale of typical values. Certainly, a value between 18 and 25 kJ/g is realistic, and an even higher one possible. This gives another factor of 1.2 – 1.7 or even more
5. Conclusions
I have shown that Basile's data proves that the red layer of his red-gray chip #13 consists of at least 87.8% combustible hydrocarbons. I further showed that, assuming the most “thermite-friendly” values of everything, at most 4.74% of the same material could be ideal thermite. I finally computed that, under the same “thermite-friendly” assumptions, thermite contributes at most 1.4% of the heat when the chip is burned. Allowing for the maximum possible amount of elemental Al given the data, but more average assumptions, it turns out that the hydrocarbon matrix provides more than 100 times the heat that thermite possibly could. The conclusions are inevitable:
The red-gray chip is not thermitic by nature – it's combustion is dominated (99-100% of the energy output) by reactions other than than the thermite reaction
Basile's data presentation in no way confirms the presence of thermite
Basile shows that the hydrocarbons in red-gray chips can burn vigorously, invalidating any claims by Harrit e.al. that the vigor of the combustion is a sign for thermite at work
If Basile, Harrit e.al. as well as other 9/11 Truthers are to be believed that “Basile's results confirm Harrit e.al.'s results”, then they must no accept that these results speak clearly against a thermitic nature of the red-gray chips
Alternatively, 9/11 Truthers should retract their stance that Basile's data “confirms” Harrit e.al.
6. References
[1] Mark Basile: 911 Dust Analysis Raises Questions. Videotaped presentation at the Porcupine Freedom Festival in Lancaster, New Hampshire on 26th June 2010, 4pm
[2] Niels H. Harrit, Jeffrey Farrer, Steven E. Jones, Kevin R. Ryan, Frank M. Legge, Daniel Farnsworth, Gregg Roberts, James R. Gourley and Bradley R. Larsen: Active Thermitic Material Discovered in Dust from the 9/11 World Trade Center Catastrophe. The Open Chemical Physics Journal, 2009, 2, 7-31
[3] Wikipedia: Energy Density. Retrieved on 2012/03/18
Newsflash!
ReplyDeleteThe chips that Basile and Harrit are nanothermite, not ordinary thermite.
Nanothermites are often mixed with carbon and silicon and other chemicals in a hybrid organic/inorganic matrix.
There is absolutely nothing suspicious about 70% of the chips being this matrix material..and that this material contributes to the energy release and the explosive nature...
These are in fact some of the signatures of superthermites!
Get a clue, get a clue....or stick to the homer simpsons of this world in the JREF forum....they do not mind if you miss little 'details' like these.
Hello Anonymous,
Deletethanks for your comment! A few remarks:
1. Neither Basile nor Harrit have proven at all that the chips are thermite of ANY kind.
2. Could you give me a reference to any publication from which I may learn that nanothermite compositiuons exist with less than 5% actual thermite in them? Thanks.
3. If you watch Basile's video closely between 41:43 and 42:00, you will see that the chip BURNS. It does NOT explode. So the organic matrix "contributes" to the flammable nature, NOT the explosive nature. Saying "contribute" is a bit misleading, by the way, when the contribution is 99-100% of the energy.
4. Again, can you link me to a publication that shows these signatures of superthermites such as
- burns slowly over a matter of seconds
- 99-100% of the energy comes from organic matrix
- <5%, and possibly 0%, of the mass is actually thermite
- >7% of the mass are other inorganic compounds
Thanks!
'Could you give me a reference to any publication from which I may learn that nanothermite compositiuons exist with less than 5% actual thermite in them? Thanks.'
ReplyDeleteI had already given you the reference in another post...but you deleted that comment..
but let me remind you again about reference 21 in Harrit et al. This report mentions organic nanothermite, that is 100% organic materials, 0% iron/al.
I await your answering at the forum
http://the911forum.freeforums.org/active-thermitic-material-in-wtc-dust-t150-150.html
Are you no-longer able to confront the forum?
I´ll even accept your challenge and prove to you that the 'official narrative' is BS.
Are you willing to break away from the JREF slim-flam and discuss some issues seriously?
Or are you deleting this as well?
Do not disappoint 'uglypig' at the forum..
Your conclusions are wrong, simply because energy density of a material, especially a nano material says nothing on their effects, when they are combined.
ReplyDeleteTo speak with you: reactions (of these materials (plural emphasized) can be quite complex.
oystein:
"Reactions will continue and can be quite complex). Wikipedia [3] lists a few organic materials and they energy density under air..."
So your critics don t fit the matter that several (and unknown) nano materials are involved.
The composition, the mixture, the size of surfaces to oxygen (nano particles!) and the arrangement of a device does it and definitely not the energy densities of individual ingredients, which are moreover not specified by you.
Therefore your conclusions are wrong.
Sorry, Johannes, I didn't check my blog for new comments since christmas or so.
DeleteI don't understand what you are saying. It sounds a bit like "it's nano! It's magic!"
You say "energy density of a material, especially a nano material says nothing on their effects, when they are combined."
BUT energy density (the theoretical maximum thereof, derived from the enthalpy of reaction) gives an absolute maximum to their "effects", namely how much energy is in fact released. And the core of my argument is: more than 98% of the chemical energy comed from organic comustion, less than 2% (probably 0%) from the thermite reaction.
You seem to imply that perhaps the nano-stuff acts as a catalyzer to the organic combustion?
Well, perhaps, even though this is neither what Harrit e.al. conclude or claim, nor is it supported by any data at all.
I loved reading this piece! Well written!
ReplyDeleteMerlen Hogg
joint
hi Oystein, can we turn it around? Try to convince me that 19 muslims disintegrated 3 sky scrapers, flew into the Pentagon, and crashed in Pennsylvania without debris. Proof it! :-)
ReplyDeleteMark
You want me to prove that "that 19 muslims disintegrated 3 sky scrapers, flew into the Pentagon, and crashed in Pennsylvania without debris"?
DeleteWhy? The bolded part is not true - why should I convince you of it?
What 19 muslims did was to crash 4 airliners. I would suggest that we first see where we have agreement. So:
Do you doubt, or agree, that an airliner was crashed into WTC1?
Do you doubt, or agree, that an airliner was crashed into WTC2?
Do you doubt, or agree, that an airliner was crashed into the Pentagon?
Do you doubt, or agree, that an airliner was crashed into a field in Pennsylvania?
These four questions leave out, for the moment, who crashed the planes and how. Just want to establish the part that it was airliners (commercial passenger planes operated in normal service by two airlines), that they were crashed (made to crash on purpose), and the four crash locations.
This comment has been removed by the author.
ReplyDeleteIf ok with you, I have added this to www.MarkBasile.org so we show both sides of the debate. Thank you.
ReplyDeleteIf ok with you, I have added this to www.MarkBasile.org so we show both sides of the debate. Thank you.
ReplyDeleteIf ok with you, I have added this to www.MarkBasile.com so we show both sides of the debate. Thank you.
ReplyDeleteI am glad and honored you did :)
Delete